

School of Public Health Professor Don Milton is leading a research study that brings both healthy people and people exposed to or diagnosed with COVID-19 into his research lab in the School of Public Health (SPH) building. The study is aimed at gathering new evidence related to how SARS-CoV-2, the virus that causes Covid-19, may spread by air. Dr. Milton’s team has carefully designed the study to limit risk to those in and around the SPH building.
It builds upon research that Dr. Milton has been conducting for 25 years related to influenza and other viruses, and relies on highly trained staff who are following the required protocols for a biosafety level 2-plus laboratory. This research could provide critical new information to inform prevention measures and advice on the use of masks. It is a great example of how the School of Public Health is fulfilling its research mission to discover new knowledge and apply it to protect and improve health.
You may have questions and concerns about what this study means for your safety when you return to work or study in the SPH building. We all want to protect ourselves and our families from any potential exposure to the virus that causes Covid-19. For this reason, we want to share with you the details about the study, the safety measures and protocols that are being strictly followed to protect the study staff and all others in the area in and around the SPH building and answers to some questions we have heard from the SPH community.
[As of June 8, 2020, the University of Maryland moved to Phase 1 for on-campus research, and as a result there are additional faculty and graduate students in the SPH building. All Phase 1 research labs have a detailed approved plan in place that specifies and adheres to recommended practices. In addition, there are required safety practices and employee check-ins for anyone who enters the building. Please visit Important Information from UMD's Division of Research on the Impact of COVID-19 on Research Activity for details.]
Where is this study taking place?
Dr. Milton’s clinical research lab for the COVID-19 study is located in the SPH building room 0117 (ground floor) closest to the lower level entrance on the Eppley Recreation Center side (west side) of the SPH building. Signs will be placed around the SPH building’s west entrance to indicate that it is a Covid-19 Study Patient Entrance. The study team is working to have a sign added to a designated parking spot in the lower parking lot (Lot SS1) to indicate that it is parking for Covid-19 study patients and a sign at Regent’s Drive and Wellness Way to direct study patients to park in Lot SS1.
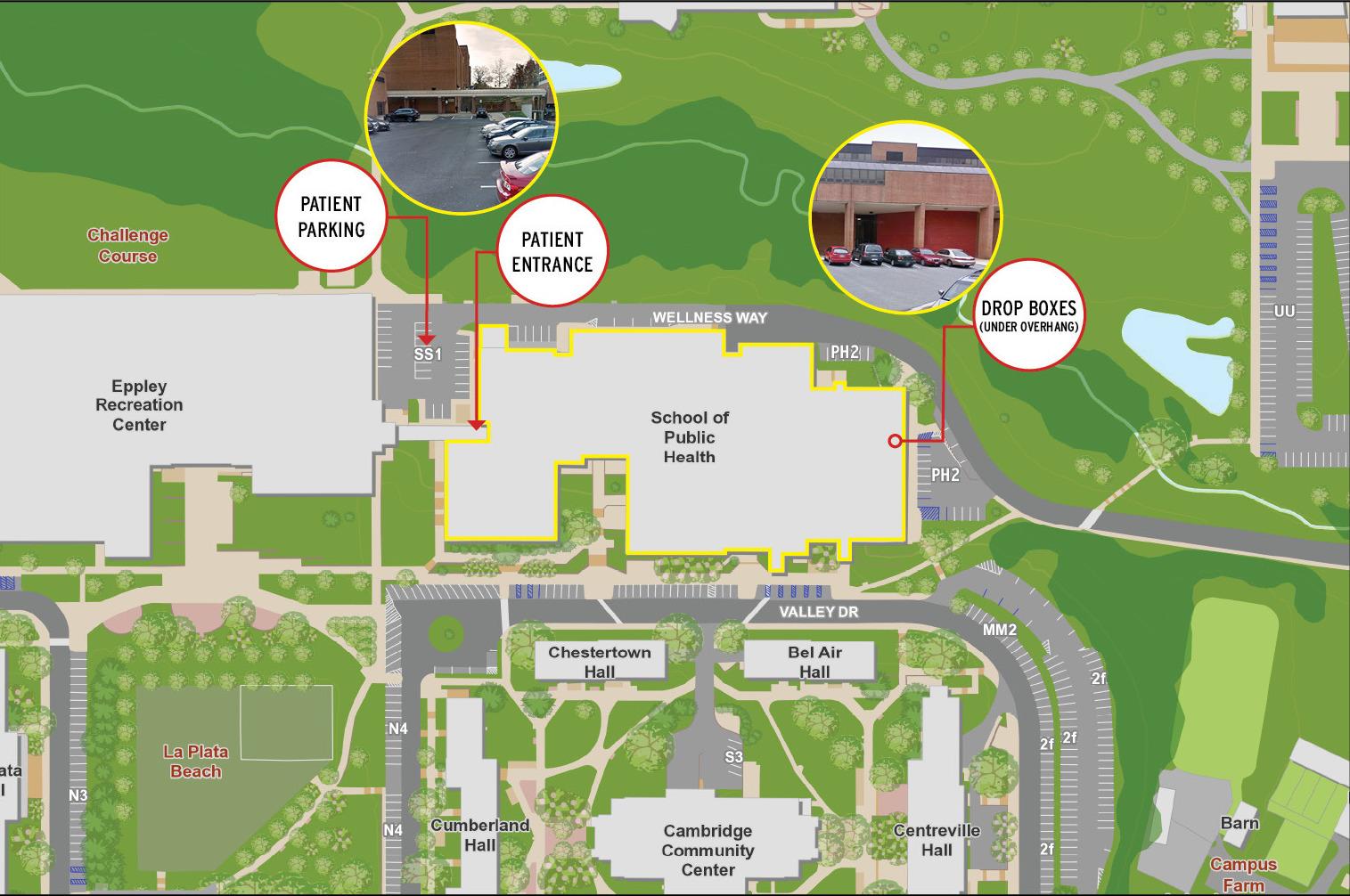
Should people avoid entering the SPH building at the west entrance?
Dr. Milton advises that people should use other entrances if possible. If you use the west entrance, he suggests people take special care to follow the social distancing and mask-wearing requirements for being on campus during phase-1 research.
What does the study involve?
Dr. Milton’s Public Health Aerobiology, Virology, and Exhaled Biomarker Laboratory (PHAB Lab) is recruiting healthy people to participate in a surveillance study and people diagnosed with Covid-19 or and their household contacts for a study to see how much virus people shed into the air from breathing and to understand what factors impact the risk of transmission. This research will gather data that will inform prevention measures and assess the effectiveness of masks in preventing the virus’s spread through the air.
Healthy surveillance study participants will be enrolled online. Participants will pick up collection tubes and drop them off at designated boxes on the east side of the SPH Building, under the overhang and next to parking lot PH2.
Participants will be instructed to pick up sampling tubes or deposit samples at indicated days/times without direct interaction with study staff and without getting closer than 6-feet from other participants while outdoors under the overhang to pick up and drop off samples.
Aerosol study participants will include people who have tested positive for Covid-19, whose illness is mild enough for them to be isolated at home, and who are within the first week since the onset of symptoms. The aerosol study participants will be enrolled online and then come to SPH for up to two in-person sampling sessions in the PHAB lab in room 0117.
Contact study participants will include close (household) contacts of known cases. The contact study participants will be enrolled online and then come to SPH every two or three days to be tested for an infection until 14 days after their last exposure to an infectious case.
Immune response study participants will include people who have recovered from COVID-19. They will enroll online and come to SPH to give blood samples approximately 3 to 4 weeks after the onset of infection, or later if not yet recovered.
How will the study manage the potential risk to those in and around the building by bringing in Covid-19 patients?
The study protocols are carefully designed to limit risk to those in and around the SPH building. Study participants must schedule an appointment in advance and call the laboratory staff from their car once on site. They are asked to park in the SS1 lower lot between the SPH building and Eppley (near the bridge to the Terrapin Trail garage). All participants will be met at their car and provided a surgical mask, and instructed to put on the surgical face mask before entering the building.
They will then be escorted from the ground floor west entrance (entrance nearest to the SPH Fitness Center) into the SPH building to the PHAB lab in room 0117 by trained clinic staff. The mask will be worn by the participant throughout the visit except during respiratory swab, saliva, and exhaled breath collection.
The lab uses the Gesundheit-II (G-II) machine to collect exhaled breath, and while the participants will not be wearing a mask for part of the time they are in the G-II, the breath collection devices are more than 90% efficient for capturing aerosol particles much smaller than the smallest respiratory viruses and the exhaust of the devices is HEPA-filtered, which eliminates the potential for infection transmission during breath sample collection.
Participants will be directed to put on their mask before exiting the G-II enclosure (the machine which captures their exhaled breath), wash their hands prior to leaving the clinic, and will wear the face mask as they are escorted by clinic staff to their car.
How will you keep the building clean and free of the virus?
SPH Room 0117
The lab in SPH Room 0117 is equipped with an upper room germicidal UV (gUV) air disinfection system and all the necessary and appropriate materials to properly collect the specimens in accordance with BSL-2 standards, including the required personal protective equipment (PPE) for both study staff and participants. The gUV light will be monitored on a regular basis to make sure it is functioning as it should. The lab does not have a ventilation system connected to the rest of the SPH building, so no air from the lab is circulated around the building via the HVAC system. The gUV system inactivates coronaviruses in the room air eliminating more than 99.9% of any airborne virus in the air every 37 seconds thus protecting room occupants as well as preventing any potential escape of virus from the room when the door is opened and closed.
Outside the SPH building:
The SPH overhang, where drop-off boxes for sample collection for the surveillance study of healthy participants will be located, is an open outdoor space and thus the risk of spreading viruses via aerosols is minimal.
Cleaning Protocols for the SPH Building
A hallway monitor from the study team will escort study participants into and out of 0117. If a participant needs to use the restroom, the hallway monitor will make sure that no one else enters while a participant is in the restroom and will spray down contact surfaces with hypochlorite and wipe down surfaces after the participant leaves.
How will you handle reporting confirmed cases of Covid-19?
All positive samples from participants who have not previously been diagnosed with SARS-CoV-2 will be reported to the appropriate health department, the University Health Center and the participant’s medical provider, if other than the UHC. The PHAB Lab turnaround for sample analysis is 12-24 hours.
Risk of research staff exposure to SARS-CoV-2 will be assessed and work restrictions and follow-up will be implemented according to the CDC guidelines. In accordance with these guidelines as of 27 March 2020, staff properly wearing a 3M Versaflo PAPR hood with face shield or similar respiratory protection, gown, and gloves will be considered to have been protected.
All study staff are required to report their temperature and symptoms everyday -- not just on days when they come to campus. Most study staff are also enrolled in the surveillance study and are tested every week. Any staff member that develops fever or "flu-like symptoms" within 14 days of sample collection or processing must contact the PI immediately by phone. Staff are issued "flu kits" containing thermometer, log sheet, pen, and surgical masks to keep at home when they join the project. Staff must use the face mask provided in the kit and self-isolate away from family / close contacts until provided direction from the PI, university Biosafety Officer, or University Health Center. Further evaluation, treatment, and decisions about testing and notification of county and state health departments will be made consistent with current Maryland Department of Health guidance.
COVID-19 information from CDC.
Who are the people involved with this study?
The project Medical Staff consists of:
- Professor Donald Milton, MD, DrPH (PI)
- Barbara Albert, MD, an SPH employee
- Oluwasanmi Oladapo Adenaiye, MBBS, MS EHS, an SPH employee
- Rhonda Washington-Lewis, an SPH employee with 25 years of clinical research experience
- Yi Esparza, RN, an SPH employee
- Jianyu Lai, BMed, MPH, an SPH doctoral student
Research staff and assistants who have completed all required clinic, IRB, and ESSR training will assist with exhaled breath sample collection (G-II) under the supervision of the Project Engineer (Somayeh Youssefi, PhD), the PI, or the Medical Staff.
The G-II operations staff include:
- Jacob Paul Bueno de Mesquita, PhD
- Somayeh Youssefi, PhD
- Dewansh Rastogi, graduate student in the SPH